Summative Evaluation / Human Factors Validation
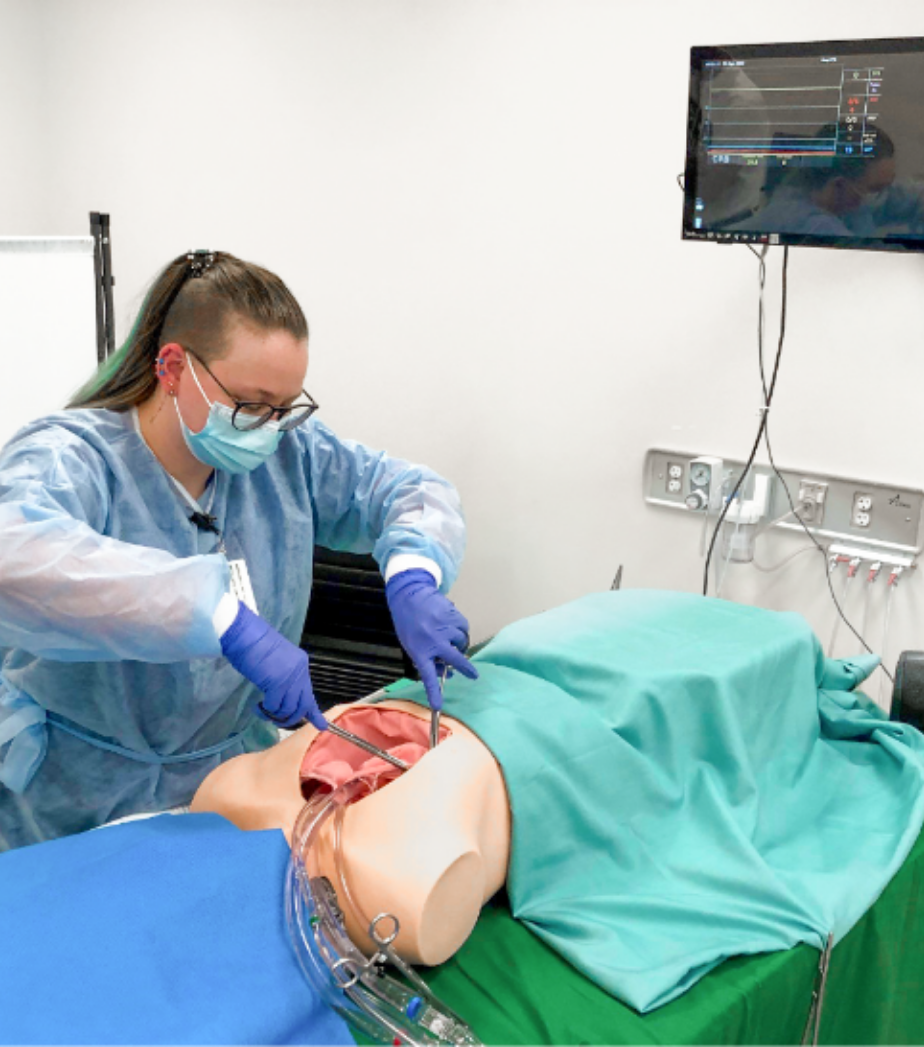
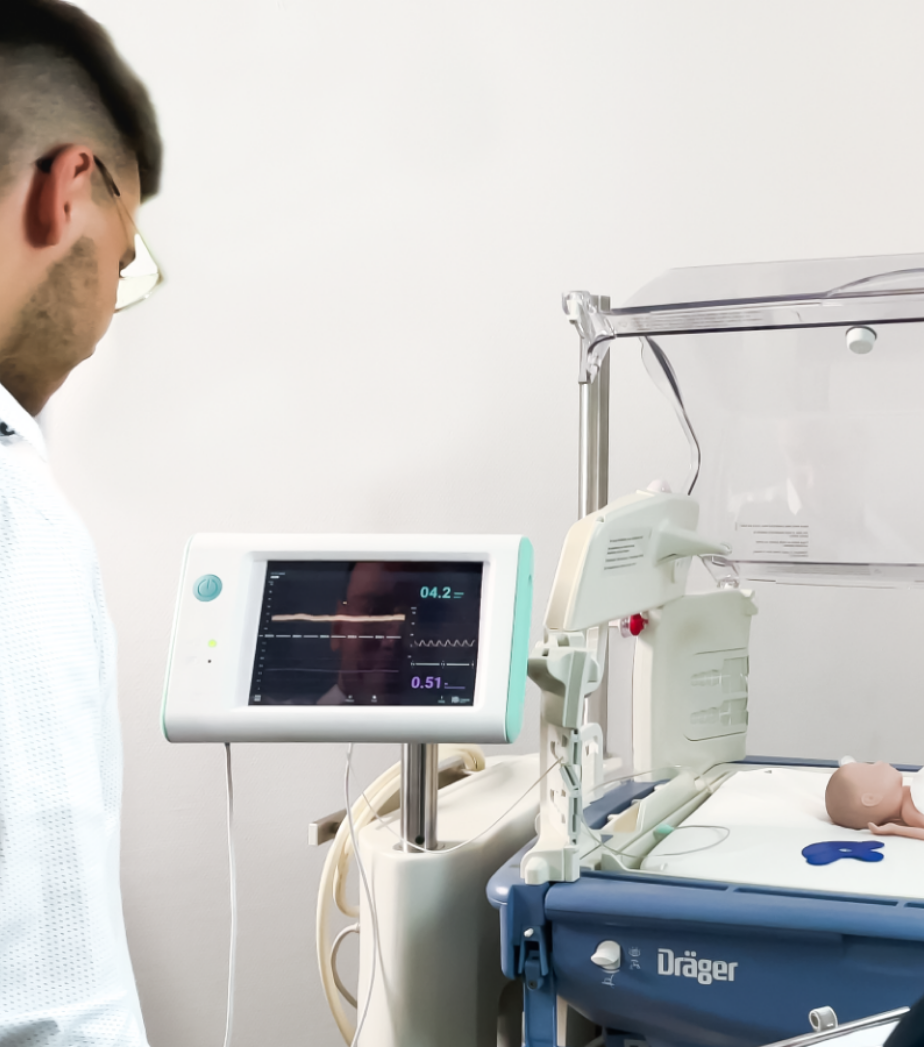
Wir unterstützen Sie bei der summativen Evaluation für den europäischen und US-amerikanischen Markt. Von der Rekrutierung der Teilnehmer bis zur erfolgreichen Durchführung sind Sie bei uns in sicheren Händen. In unseren UX-Medical-Laboren lässt sich jede Nutzungsumgebung problemlos simulieren.
Ihnen steht hier High-End Equipment (wie ein Patientensimulator oder ein kompletter gestaltbarer OP) zur Verfügung. Für Livestreams und Aufnahmen der Durchführung bieten wir Ihnen ausreichend hochauflösende Kameras.
01
Kick-off
In unserem Kick-off planen wir gemeinsam das Projekt und stellen sicher, dass alle Kernfragen und Erwartungen festgelegt sind. Hier lernen wir auch Ihr Produkt sowie Ihre Hazard-related Use Scenarios und die Risikoanalyse kennen. Zusammen legen wir einen finalen Zeitplan fest und stellen sicher, dass das Projekt genau nach Ihren Wünschen verläuft.


02
Rekrutierung der Teilnehmer
Wir rekrutieren für Sie die richtigen Teilnehmer für Ihre Studie. Egal, ob Patient oder Health Care Professional – wir greifen auf eine große Datenbank aus Nutzern zurück. Die Kriterien für die Rekrutierung legen wir dabei in Absprache mit Ihnen fest.
03
Planen der summativen Evaluation
Wir übernehmen für Sie die komplette Planung der summativen Evaluation. Das gilt von der Erstellung des Studienleitfadens bis zur Vorbereitung unseres Usability Labors mit Ihrem speziellen Testaufbau.


04
Wenn notwendig – Training der Nutzer
Gemäß Ihrer Instructions for Use oder Ihrem Trainingsmaterial schulen wir die Nutzer für Ihre Studie. Natürlich beachten wir hierbei auch eine gegebenenfalls vorhandene Decay-Zeit, also den realistischen Abstand zwischen Schulung und Nutzung.
05
Durchführung der summativen Evaluation
Wir führen für Sie die Tests durch, nach Leitfaden und inklusive Root Cause Analyse versteht sich. Nach einem ersten Test sammeln wir Ihr Feedback ein, um Anpassungen am Ablauf zu integrieren, um dann mit den restlichen Probanden zu testen. So gehen wir sicher, dass die Durchführung nach Ihren Wünschen verläuft. Gerne können Sie auch live dabei sein – vor Ort oder remote.
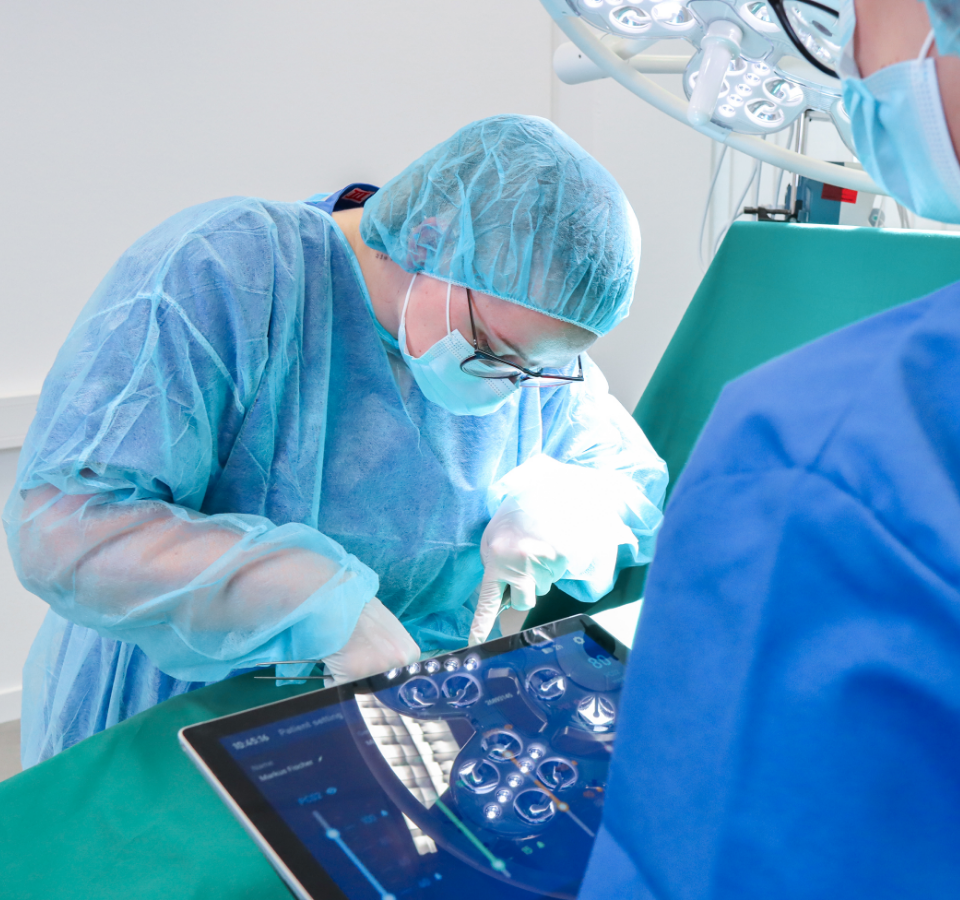

06
Auswertung & Dokumentation
Wir werten die summative Evaluation für Sie aus. Sie erhalten alle Ergebnisse Ihrer summativen Evaluation als Written Report. Die Ergebnisse der Root Cause Analyse sind hier ebenfalls enthalten. Dies ist besonders wichtig für Ihre Argumentation gegenüber der Benannten Stelle.
07
Ergebnispräsentation
Wir stellen Ihnen zum Abschluss des Projekts die Ergebnisse gesammelt vor und übergeben Ihnen den Test Report. Hiermit haben Sie den Nachweis, dass Ihr Produkt ohne inakzeptable Risiken durch die Nutzer zu verwenden ist..

Unsere Referenzen
Lassen Sie uns beginnen!
Lust auf eine schnelle, zielgerichtete und pragmatische Zusammenarbeit? Melden Sie sich und wir finden gemeinsam heraus, wie wir Sie unterstützen können.